Renal Physiology — MCQs

On this page
What is the correct formula for calculating the Glomerular Filtration Rate (GFR)?
Which of the following statements is true regarding the given cystometrogram?
Impaired function of Aquaporin results in
Vasopressin acts through which aquaporin channels in the collecting duct?
Which of the following is NOT a component responsible for the counter current mechanism in the kidney?
What is the physiological response of the kidney during shock?
Tubuloglomerular feedback control is useful for which one of the following?
Tubuloglomerular feedback is for regulation of?
Which carrier pump is responsible for transporting solutes in the thick ascending limb of the loop of Henle?
Mechanism of secretion of ammonia in distal tubule is?
Renal Physiology Indian Medical PG Practice Questions and MCQs
Question 501: What is the correct formula for calculating the Glomerular Filtration Rate (GFR)?
- A. GFR (mL/min/1.73 m^2) = 175 x (Scr)^-1.254 x (Age)^-0.203 x (0.742 if female)
- B. GFR (mL/min/1.73 m^2) = 175 x (Scr)^-1.154 x (Age)^-0.203 x (0.742 if female)
- C. GFR (mL/min/1.73 m^2) = 186 x (Scr)^-1.154 x (Age)^-0.203 x (0.742 if female) (Correct Answer)
- D. GFR (mL/min/1.73 m^2) = 175 x (Scr)^-1.154 x (Age)^-0.303 x (0.742 if female)
Explanation: ***GFR (mL/min/1.73 m^2) = 186 x (Scr)^-1.154 x (Age)^-0.203 x (0.742 if female)*** - This represents the **four-variable MDRD (Modification of Diet in Renal Disease) Study equation**, which is the most widely used formula for estimating GFR in clinical practice. - The coefficient **186** is standard for the MDRD equation when serum creatinine is measured using **IDMS-traceable methods**. - The formula uses **serum creatinine (Scr) in mg/dL**, **age in years**, and applies a correction factor of **0.742 for females**. - This equation is commonly taught in medical curricula and used for **chronic kidney disease (CKD) staging**. *GFR (mL/min/1.73 m^2) = 175 x (Scr)^-1.154 x (Age)^-0.203 x (0.742 if female)* - This formula uses a coefficient of **175**, which was used in earlier versions of the MDRD equation with different creatinine calibration methods. - While mathematically similar to the MDRD formula, the **186 coefficient is the standard** for modern IDMS-calibrated creatinine assays. - Using 175 instead of 186 would result in **underestimation of GFR** by approximately 6%. *GFR (mL/min/1.73 m^2) = 175 x (Scr)^-1.254 x (Age)^-0.203 x (0.742 if female)* - This option has an incorrect exponent for **serum creatinine (Scr)**; the correct exponent in the MDRD formula is **-1.154**, not -1.254. - Errors in the creatinine exponent would drastically alter the calculated GFR and lead to **inaccurate kidney function assessment**. *GFR (mL/min/1.73 m^2) = 175 x (Scr)^-1.154 x (Age)^-0.303 x (0.742 if female)* - This option incorrectly uses an exponent of **-0.303 for age**, whereas the correct exponent for age in the MDRD equation is **-0.203**. - An incorrect age exponent would lead to **misestimation of GFR**, particularly affecting older patients where age-related decline in kidney function is clinically significant.
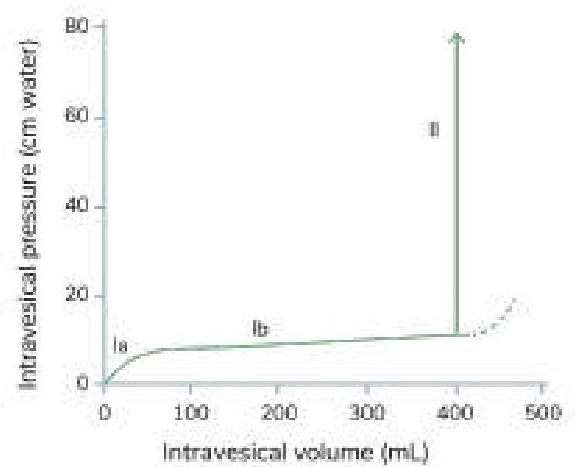
Question 502: Which of the following statements is true regarding the given cystometrogram?
- A. Segment Ib reflects the bladder's ability to accommodate increasing volume without a significant rise in pressure. (Correct Answer)
- B. Micturition occurs in segment II; failure to micturate is not characteristic of this segment.
- C. Segment Ia represents the initial phase of bladder filling, not residual urine.
- D. The dotted line represents a reference point, not the occurrence of micturition.
Explanation: ***Segment Ib reflects the bladder's ability to accommodate increasing volume without a significant rise in pressure.*** - Segment Ib typically represents the **storage phase** of the bladder, where the detrusor muscle relaxes allowing for significant increases in volume with only a small increase in intravesical pressure. - This property is known as **compliance** and is crucial for normal bladder function, preventing premature urgency and high-pressure storage. - This is the **most clinically significant characteristic** that defines normal bladder function. *Micturition occurs in segment II; failure to micturate is not characteristic of this segment.* - While this statement is technically true, segment II represents the **micturition phase** with rapid rise in intravesical pressure as the detrusor contracts. - The statement is correct but less specific about the key functional property being tested in a cystometrogram. *Segment Ia represents the initial phase of bladder filling, not residual urine.* - This statement is also technically true; segment Ia represents the **initial filling phase** where the bladder begins to distend from baseline. - However, this is a basic anatomical description rather than a functionally significant characteristic. *The dotted line represents a reference point, not the occurrence of micturition.* - The dotted line typically shows a further increase in intravesical pressure, indicating **maximal pressure** the bladder can withstand or continued voiding attempt against resistance. - Segment II (solid line with rapid pressure increase) is where micturition actively occurs, making this a true but less significant observation.
Question 503: Impaired function of Aquaporin results in
- A. Nephrogenic DI (Correct Answer)
- B. Liddle syndrome
- C. Cystic fibrosis
- D. Bartter syndrome
Explanation: ***Nephrogenic DI*** - **Nephrogenic Diabetes Insipidus (DI)** results from the kidneys' inability to respond to **vasopressin (ADH)** due to defects in **V2 receptors** or, more commonly, mutations in **Aquaporin-2** (AQP2) water channels. - This leads to the excretion of large volumes of dilute urine, as water cannot be reabsorbed by the collecting ducts even when ADH levels are adequate. *Liddle syndrome* - This is an **autosomal dominant** disorder characterized by unregulated activation of the **epithelial sodium channel (ENaC)** in the collecting tubules. - It leads to increased sodium reabsorption, hypertension, hypokalemia, and metabolic alkalosis, not directly related to aquaporin function. *Cystic fibrosis* - This is an **autosomal recessive** genetic disorder that affects cells that produce mucus, sweat, and digestive juices, primarily due to mutations in the **CFTR (cystic fibrosis transmembrane conductance regulator) gene**. - Impaired CFTR protein function leads to thick, sticky secretions that can block ducts and passages, primarily in the lungs and pancreas, and is unrelated to aquaporin function. *Bartter syndrome* - This is a group of **autosomal recessive** disorders characterized by impaired reabsorption of sodium and chloride in the **thick ascending limb of the loop of Henle**. - It leads to significant urine loss of sodium, potassium, and chloride, resulting in hypokalemia, metabolic alkalosis, hypercalciuria, and often normal to low blood pressure, not directly related to aquaporin.
Question 504: Vasopressin acts through which aquaporin channels in the collecting duct?
- A. Aquaporin 1
- B. Aquaporin 2 (Correct Answer)
- C. Aquaporin 4
- D. Aquaporin 3
Explanation: ***Aquaporin 2*** - Vasopressin (ADH) stimulates the insertion of **Aquaporin 2 (AQP2)** channels into the apical membrane of collecting duct cells, increasing water reabsorption. - This process is crucial for the kidney's ability to concentrate urine and maintain **water balance**. *Aquaporin 1* - **Aquaporin 1 (AQP1)** is predominantly found in the proximal tubules and descending limb of the loop of Henle, where **constitutive water reabsorption** occurs, independent of vasopressin. - It plays a role in bulk water reabsorption rather than regulated fine-tuning. *Aquaporin 3* - **Aquaporin 3 (AQP3)** is located on the **basolateral membrane** of collecting duct cells, facilitating the exit of water from the cell into the interstitial fluid. - While essential for water movement, its insertion into the membrane is **not directly regulated by vasopressin** in the same way as AQP2. *Aquaporin 4* - **Aquaporin 4 (AQP4)** is also found on the **basolateral membrane** of collecting duct cells and in other tissues like the brain. - Similar to AQP3, it allows water to leave the cell but is not the primary target for vasopressin-mediated regulation of water permeability.
Question 505: Which of the following is NOT a component responsible for the counter current mechanism in the kidney?
- A. Sodium outflow in thick ascending limb
- B. Flow of tubular fluid from PCT to DCT (Correct Answer)
- C. Sodium outflow in thin ascending limb
- D. Water outflow in thin descending limb
Explanation: ***Flow of tubular fluid from PCT to DCT*** - While tubular fluid flow through the nephron is necessary for overall kidney function, it is **NOT a specific component of the countercurrent mechanism** itself. - The countercurrent mechanism specifically refers to the **countercurrent multiplier system** in the **Loop of Henle**, which creates and maintains the medullary osmotic gradient. - The flow from PCT to DCT is simply the sequential passage of fluid through nephron segments, not a specific mechanism for creating the hypertonic medullary interstitium. *Sodium outflow in thick ascending limb* - The **thick ascending limb** actively transports **Na+, K+, and Cl- ions** (via Na-K-2Cl cotransporter) out of the tubule into the medullary interstitium. - This segment is **impermeable to water**, allowing solute removal without water, creating a **dilute tubular fluid** and **hypertonic interstitium**. - This is the **primary active component** of the countercurrent multiplier mechanism. *Water outflow in thin descending limb* - The **thin descending limb** is highly permeable to **water** but relatively impermeable to solutes. - As tubular fluid descends into the hypertonic medulla, water passively moves out, **concentrating the tubular fluid**. - This passive water reabsorption is essential for the countercurrent mechanism. *Sodium outflow in thin ascending limb* - The **thin ascending limb** is permeable to Na+ and Cl-, allowing **passive diffusion** of these ions into the medullary interstitium. - Though less significant than the active transport in the thick ascending limb, this passive sodium outflow **contributes to the osmotic gradient** in the medulla. - This segment is also **impermeable to water**, preventing water reabsorption while allowing solute loss.
Question 506: What is the physiological response of the kidney during shock?
- A. GFR decreases
- B. Perfusion of kidney decreases
- C. Afferent arteriole resistance increases
- D. Renal blood flow decreases (Correct Answer)
Explanation: ***Renal blood flow decreases*** - During shock, the **primary and most fundamental** physiological change affecting the kidney is a marked **reduction in renal blood flow (RBF)**. - Shock triggers intense **sympathetic activation** and **renin-angiotensin system (RAS) activation**, causing preferential **vasoconstriction** of renal vessels to redirect blood to vital organs (brain, heart). - RBF can drop to as low as **20-30% of normal** in severe shock, making this the hallmark renal response. - This reduction in RBF is the **upstream event** that triggers all other renal changes during shock. *Perfusion of kidney decreases* - While technically correct, "decreased perfusion" is **essentially synonymous** with decreased blood flow in this context. - The term "renal blood flow" is the **standard physiological terminology** used in medical literature to describe this phenomenon, making it the more precise answer. *Afferent arteriole resistance increases* - This is a **mechanism** by which RBF decreases, not the overall response itself. - Increased afferent arteriolar resistance is **secondary** to sympathetic activation and angiotensin II effects during shock. - It describes the "how" rather than the "what" of the kidney's response. *GFR decreases* - GFR reduction is a **consequence** of decreased RBF and increased afferent arteriolar resistance. - While clinically important (oliguria/acute kidney injury), it's a **downstream effect** rather than the primary physiological response. - The relationship: ↓RBF → ↓Glomerular hydrostatic pressure → ↓GFR
Question 507: Tubuloglomerular feedback control is useful for which one of the following?
- A. GFR (Correct Answer)
- B. Plasma sodium
- C. Plasma volume
- D. Determining tubular secretion
Explanation: ***GFR*** - **Tubuloglomerular feedback (TGF)** is a critical autoregulatory mechanism that maintains a relatively constant **glomerular filtration rate (GFR)** despite fluctuations in arterial blood pressure. - The **macula densa** cells at the end of the thick ascending limb of the loop of Henle sense the **volume** and **sodium chloride concentration** of the tubular fluid and release paracrine factors to adjust afferent arteriolar resistance. *Plasma sodium* - While TGF senses the **sodium chloride concentration** in the filtrate, its primary role is to regulate GFR, not directly control systemic plasma sodium levels. - Plasma sodium is primarily regulated by hormones like **ADH** and **aldosterone**, which influence water reabsorption and sodium excretion. *Plasma volume* - **Plasma volume** is regulated predominantly by hormonal mechanisms (e.g., **renin-angiotensin-aldosterone system**, **ADH**, **ANP**) and control over overall sodium and water balance, rather than by the acute, intrinsic GFR regulation of TGF. - Changes in plasma volume can indirectly affect GFR, but TGF is not the direct control mechanism for plasma volume itself. *Determining tubular secretion* - **Tubular secretion** is the process by which solutes are actively transported from the peritubular capillaries into the tubular lumen. - TGF influences **glomerular filtration**, not directly the rates of tubular secretion, which are regulated by specific transport proteins and physiological needs.
Question 508: Tubuloglomerular feedback is for regulation of?
- A. BP
- B. Blood volume
- C. Na+ reabsorption
- D. Renal blood flow (Correct Answer)
Explanation: ***Renal blood flow*** - **Tubuloglomerular feedback (TGF)** is a key mechanism that helps to tightly regulate **renal blood flow** and **glomerular filtration rate (GFR)** by sensing tubular fluid composition. - This mechanism involves the **macula densa** cells in the distal tubule sensing changes in **sodium chloride (NaCl) delivery**, which then signals the afferent arteriole to adjust its caliber. *BP* - While renal blood flow ultimately influences **blood pressure (BP)**, tubuloglomerular feedback is primarily focused on **local autoregulation** within the kidney, not systemic BP control. - Systemic BP is regulated by much broader mechanisms involving the **renin-angiotensin-aldosterone system** and **autonomic nervous system**. *Blood volume* - **Blood volume** is primarily regulated by hormones like **ADH (vasopressin)** and **aldosterone**, which influence water and sodium reabsorption in the collecting ducts and other parts of the nephron. - Although renal function impacts blood volume, tubuloglomerular feedback's direct role is not in regulating the overall volume of blood. *Na+ reabsorption* - While the macula densa senses **Na+ delivery** to the distal tubule and this influences GFR, the primary role of tubuloglomerular feedback is not to modulate the total amount of **Na+ reabsorbed** throughout the nephron. - Na+ reabsorption is regulated by multiple factors and occurs extensively in the **proximal tubule**, **loop of Henle**, and **distal tubule**, often under hormonal control.
Question 509: Which carrier pump is responsible for transporting solutes in the thick ascending limb of the loop of Henle?
- A. NaCl cotransporter
- B. Na+-H+ exchanger
- C. Na+-K+ exchanger
- D. Sodium-potassium-chloride cotransporter (Correct Answer)
Explanation: * **Sodium-potassium-chloride cotransporter.** * This transporter, specifically the **Na+-K+-2Cl- cotransporter (NKCC2)**, is highly expressed in the apical membrane of the thick ascending limb. * It actively reabsorbs **sodium, potassium, and chloride ions** from the filtrate, contributing significantly to the medullary interstitial osmotic gradient. * *NaCl- cotransporter* * The **NaCl cotransporter (NCC)** is primarily found in the **distal convoluted tubule**, not the thick ascending limb. * It reabsorbs sodium and chloride in a 1:1 ratio and is the target of thiazide diuretics. * *Na+-H+ exchanger* * The **Na+-H+ exchanger (NHE3)** is predominantly located in the **proximal tubule** where it plays a crucial role in bicarbonate reabsorption and acid-base balance. * While some NHE activity exists in other nephron segments, it is not the primary carrier in the thick ascending limb. * *Na+-K+ exchanger* * The **Na+-K+ exchanger** or **Na+/K+-ATPase pump** is located on the basolateral membrane of most renal tubular cells, including the thick ascending limb. * Its main function is to maintain the electrochemical gradient by pumping **sodium out of the cell** and potassium into the cell, which indirectly drives other transporters but is not the apical cotransporter responsible for initial solute reabsorption in the thick ascending limb.
Question 510: Mechanism of secretion of ammonia in distal tubule is?
- A. Primary active transport
- B. Symport
- C. Antiport
- D. Passive diffusion (Correct Answer)
Explanation: ***Passive diffusion*** - Ammonia (NH3) is a **lipid-soluble molecule** that can readily cross cell membranes, including those of the distal tubule and collecting duct, down its **concentration gradient**. - This process is crucial for regulating **acid-base balance**, as NH3 traps H+ ions to form NH4+, which is then excreted. *Primary active transport* - This mechanism involves the direct use of **ATP hydrolysis** to move ions against their concentration gradient, which is not the primary way ammonia is secreted in the distal tubule. - While NH4+ can be secreted via active transport in some segments (e.g., substituting for K+ on the Na-K-2Cl cotransporter in the thick ascending limb), free ammonia diffusion is distinct. *Symport* - **Symport** involves the co-transport of two or more different molecules or ions in the same direction across a cell membrane, powered by an electrochemical gradient. - This mechanism is not typically involved in the secretion of uncharged, lipid-soluble ammonia. *Antiport* - **Antiport** is a type of coupled transport where two different ions or molecules move in opposite directions across a membrane. - While antiport systems are essential for various renal functions (e.g., Na+/H+ exchanger), they are not the primary mechanism for the secretion of free ammonia in the distal tubule.
Practice by Chapter
Renal Blood Flow and Glomerular Filtration
Practice Questions
Tubular Reabsorption and Secretion
Practice Questions
Concentration and Dilution of Urine
Practice Questions
Acid-Base Regulation by the Kidneys
Practice Questions
Sodium and Water Balance
Practice Questions
Potassium Regulation
Practice Questions
Calcium and Phosphate Handling
Practice Questions
Micturition Physiology
Practice Questions
Renal Function Tests
Practice Questions
Integrative Responses to Fluid Challenges
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Start For Free