Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Practice Questions and MCQs
Practice Indian Medical PG questions for Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve. These multiple choice questions (MCQs) cover important concepts and help you prepare for your exams.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Question 1: Which factor predominantly influences the rightward shift of the oxygen dissociation curve?
- A. pH (Bohr effect)
- B. 2,3-Bisphosphoglycerate (2,3-BPG) (Correct Answer)
- C. Temperature increase
- D. Carbon monoxide levels
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Explanation: ***2,3-Bisphosphoglycerate (2,3-BPG)***
- **2,3-BPG** is an organic phosphate found in **red blood cells** that serves as the **predominant regulator** of oxygen-hemoglobin affinity under physiological conditions.
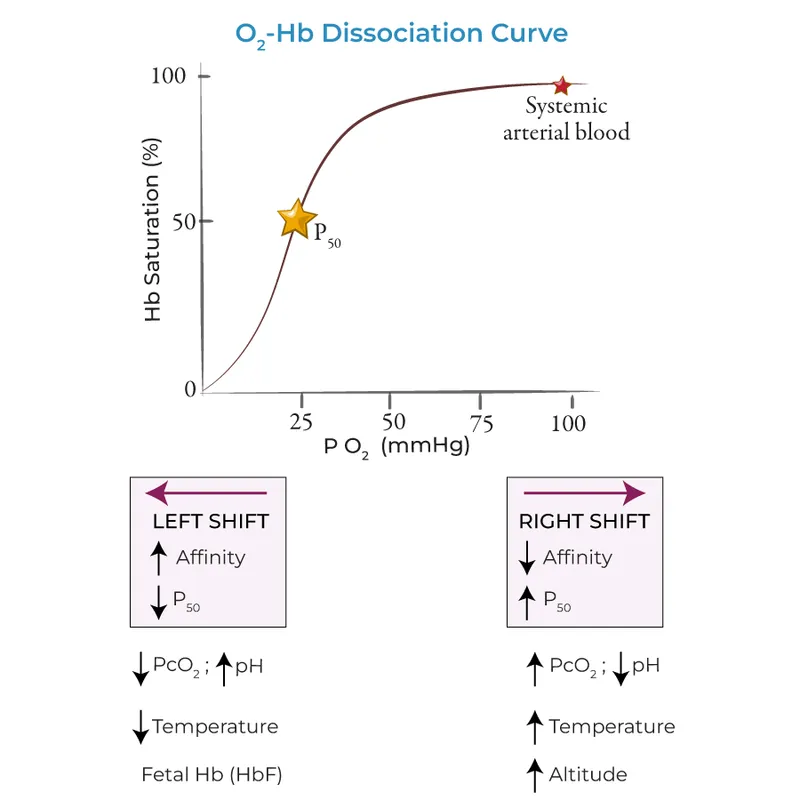
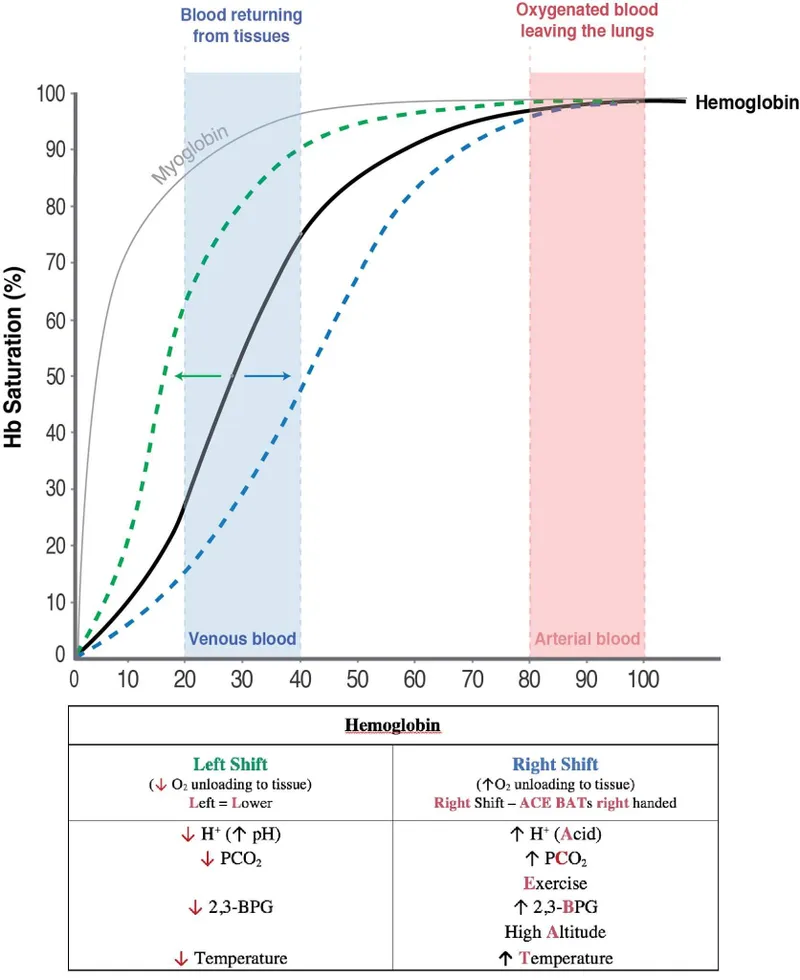
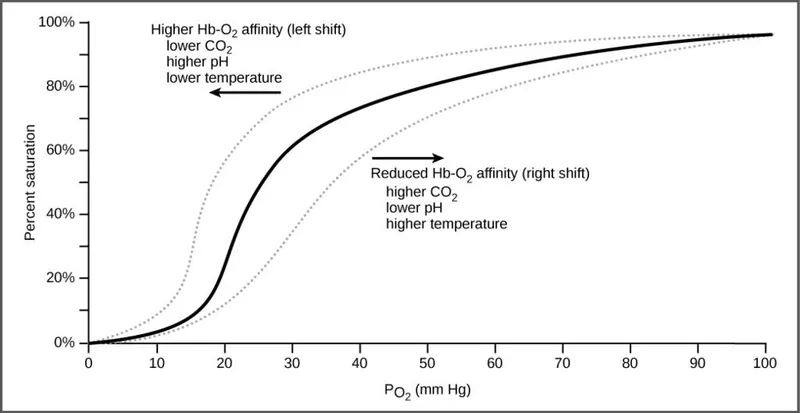
- An increase in **2,3-BPG** levels binds to the **beta chains of deoxyhemoglobin**, stabilizing the T (tense) state and reducing hemoglobin's affinity for oxygen, thereby shifting the curve to the right and facilitating **oxygen release** to tissues.
- **2,3-BPG** is especially important in **chronic adaptations** to hypoxia (high altitude, chronic lung disease, anemia) and is the **primary mechanism** for sustained alterations in oxygen delivery.
- Normal RBC 2,3-BPG concentration is approximately equal to hemoglobin concentration, making it a **quantitatively significant** regulatory factor.
*pH (Bohr effect)*
- A decrease in blood **pH** (increased acidity) due to higher **CO2** and **H+** concentrations also shifts the oxygen dissociation curve to the right via the **Bohr effect**.
- While physiologically important for **acute regulation** in metabolically active tissues, the Bohr effect operates in conjunction with other factors rather than as the predominant standalone regulator.
- The effect is mediated by **protonation of histidine residues** on hemoglobin, causing conformational changes that reduce oxygen affinity.
*Temperature increase*
- An increase in **temperature** reduces hemoglobin's affinity for oxygen, shifting the oxygen dissociation curve to the right.
- This effect is vital for **oxygen delivery** to actively metabolizing tissues (which generate heat), but is generally a **secondary factor** compared to 2,3-BPG in terms of overall regulation.
- The temperature effect is more situational, occurring primarily in tissues with elevated metabolic activity.
*Carbon monoxide levels*
- **Carbon monoxide (CO)** causes a **leftward shift** of the oxygen dissociation curve, not a rightward shift.
- CO binds to hemoglobin with 200-250 times greater affinity than oxygen, forming **carboxyhemoglobin** (COHb).
- This not only reduces oxygen-carrying capacity but also **increases hemoglobin's affinity** for the remaining oxygen, making it harder to release oxygen to tissues.
- CO poisoning is therefore dangerous both because it displaces oxygen and because it impairs oxygen delivery through leftward shift.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Question 2: The oxygen-hemoglobin dissociation curve is sigmoid because
- A. Binding of one oxygen molecule decreases the affinity of binding other O2 molecules
- B. Oxygen affinity of Hemoglobin decreases when the pH of blood falls
- C. Binding of oxygen to Hemoglobin reduces the affinity of Hb for CO2
- D. Binding of one oxygen molecule increases the affinity of binding other O2 molecules (Correct Answer)
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Explanation: ***Binding of one oxygen molecule increases the affinity of binding other O2 molecules***
- The **sigmoid shape** of the oxygen-hemoglobin dissociation curve reflects the cooperative binding of oxygen. When one oxygen molecule binds to a heme unit in hemoglobin, it causes a conformational change that increases the affinity of the remaining heme units for oxygen.
- This **cooperative binding** means that at low partial pressures of oxygen, very little oxygen binds to hemoglobin. However, once a few oxygen molecules bind, subsequent binding occurs much more readily and steeply, leading to the characteristic 'S' shape.
*Binding of one oxygen molecule decreases the affinity of binding other O2 molecules*
- This statement is incorrect as it describes **negative cooperativity**, which is the opposite of what occurs with oxygen and hemoglobin.
- Decreased affinity after initial binding would lead to a **hyperbolic (rectangular)** curve rather than a sigmoid one, similar to myoglobin's oxygen binding curve.
*Oxygen affinity of Hemoglobin decreases when the pH of blood falls*
- This describes the **Bohr effect**, where a decrease in pH (acidosis) or an increase in CO2 shifts the curve to the right, indicating reduced oxygen affinity and enhanced oxygen release to tissues.
- While this is an important physiological phenomenon, it explains the **shift** of the curve rather than its inherent **sigmoid shape**.
*Binding of oxygen to Hemoglobin reduces the affinity of Hb for CO2*
- This phenomenon is known as the **Haldane effect**, where oxygen binding promotes the release of CO2 from hemoglobin in the lungs.
- The Haldane effect is another crucial aspect of hemoglobin function but does not explain the **sigmoid shape** of the oxygen-hemoglobin dissociation curve itself.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Question 3: Which poison shows cherry red discoloration of blood but normal PaO2 on blood gas analysis?
- A. Cyanide
- B. Hydrogen sulfide
- C. Carbon monoxide (Correct Answer)
- D. Nitrites
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Explanation: ***Carbon monoxide***
- **Carbon monoxide (CO)** binds to **hemoglobin** with a much higher affinity than oxygen, forming **carboxyhemoglobin**. This complex is bright red, causing the characteristic **cherry-red discoloration of blood** and skin.
- Despite the impaired oxygen delivery, the partial pressure of dissolved oxygen in the blood (**PaO2**) remains normal because CO poisoning affects oxygen binding to hemoglobin rather than the amount of oxygen dissolved in plasma.
*Cyanide*
- **Cyanide** inhibits **cytochrome c oxidase**, impairing cellular oxygen utilization and leading to **lactic acidosis** and cellular hypoxia.
- While it can cause cellular hypoxia, it does not typically produce cherry-red discoloration and usually results in an **arteriovenous oxygen difference** that is small as tissues cannot extract oxygen from the blood effectively.
*Hydrogen sulfide*
- **Hydrogen sulfide (H2S)** also inhibits **cytochrome c oxidase**, leading to cellular hypoxia similar to cyanide.
- Although it can cause a "rotten egg" smell and rapid collapse, it does not typically produce the characteristic **cherry-red discoloration** of blood.
*Nitrites*
- **Nitrites** (and other oxidizing agents) cause **methemoglobinemia**, where the iron in hemoglobin is oxidized from the ferrous (Fe2+) to the ferric (Fe3+) state, which cannot bind oxygen.
- This condition causes the blood to appear **chocolate brown** or **bluish-gray**, not cherry-red, and can lead to a **functional anemia** despite normal PaO2.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Question 4: Which of the following laboratory findings is most consistent with a diagnosis of carbon monoxide poisoning?
- A. Increased PaCO2 and decreased pH
- B. Decreased PaO2 with normal oxygen saturation
- C. Normal PaO2 with decreased oxygen saturation (Correct Answer)
- D. Decreased PaCO2 with normal PaO2
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Explanation: ***Normal PaO2 with decreased oxygen saturation***
- Carbon monoxide (CO) binds to hemoglobin with an affinity 200-250 times greater than oxygen, forming **carboxyhemoglobin (COHb)** [2]. This reduces the **oxygen-carrying capacity** of the blood and shifts the oxygen dissociation curve to the left, but it does **not affect the partial pressure of oxygen (PaO2)** dissolved in the plasma [1].
- The pulse oximeter, which typically measures oxygen saturation, will show a falsely high reading because it cannot differentiate between oxyhemoglobin and carboxyhemoglobin, but actual **oxygen saturation is decreased**.
*Increased PaCO2 and decreased pH*
- This pattern suggests **respiratory acidosis**, which is not a direct or primary finding of carbon monoxide poisoning.
- While severe CO poisoning can lead to lactic acidosis, an increase in PaCO2 points to impaired ventilation, not specifically CO toxicity [3].
*Decreased PaO2 with normal oxygen saturation*
- A decreased PaO2 with normal oxygen saturation is a contradictory finding and not physiologically consistent, as oxygen saturation is directly dependent on PaO2.
- This pattern would indicate a measurement error or a highly unusual physiological state, neither of which is characteristic of CO poisoning.
*Decreased PaCO2 with normal PaO2*
- This suggests **respiratory alkalosis**, often due to hyperventilation.
- While patients with CO poisoning may hyperventilate due to hypoxia, this ABG pattern is not the defining laboratory finding for CO poisoning, and **PaO2 would remain normal** until very late stages.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Question 5: How many Fe²⁺ atoms are present in one molecule of hemoglobin (Hb)?
- A. One Fe²⁺ atom
- B. Two Fe²⁺ atoms
- C. Four Fe²⁺ atoms (Correct Answer)
- D. Eight Fe²⁺ atoms
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Explanation: ***Four Fe²⁺ atoms***
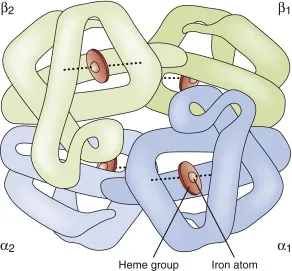
- A single molecule of **hemoglobin** is composed of **four globin chains**, each containing one **heme group**.
- Each **heme group** in hemoglobin contains one central **ferrous iron (Fe²⁺) atom**, allowing for the binding of one oxygen molecule per heme group.
*One Fe²⁺ atom*
- This is incorrect because hemoglobin is a **tetramer**, meaning it has multiple subunits.
- Only one heme group (and thus one Fe²⁺ atom) is present in **myoglobin**, which is a single polypeptide chain, not hemoglobin.
*Two Fe²⁺ atoms*
- This is incorrect as it does not account for the **tetrameric structure** of adult hemoglobin.
- While some developmental forms of hemoglobin could be considered to have two alpha and two beta chains, each still has its own heme group.
*Eight Fe²⁺ atoms*
- This is incorrect as it would imply two Fe²⁺ atoms per heme group or multiple heme groups per globin chain.
- The 1:1 ratio of heme group to Fe²⁺ atom and globin chain to heme group is fundamental to hemoglobin structure.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Question 6: The daily production of hydrogen ions from CO2 is primarily buffered by which of the following?
- A. Red blood cell bicarbonate
- B. Extracellular bicarbonate
- C. Plasma proteins
- D. Red blood cell hemoglobin (Correct Answer)
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Explanation: ***Red blood cell hemoglobin***
- **Hemoglobin is the primary buffer** for the massive daily acid load from CO2 (approximately 12,500 mEq H+ per day).
- CO2 diffuses into RBCs where **carbonic anhydrase** rapidly catalyzes: CO2 + H2O → H2CO3 → H+ + HCO3-.
- **Deoxygenated hemoglobin** has a higher affinity for H+ than oxygenated hemoglobin (reduced hemoglobin is a weaker acid, thus better H+ acceptor).
- This buffering is crucial for CO2 transport: **Hb + H+ → HHb**, preventing significant pH changes despite huge CO2 production.
- The bicarbonate produced is then transported out via the **chloride shift** to maintain electrical neutrality.
*Extracellular bicarbonate*
- While the bicarbonate buffer system is quantitatively the largest extracellular buffer, it is **NOT the primary buffer for CO2-derived H+**.
- The extracellular HCO3-/CO2 system primarily buffers **metabolic (non-volatile) acids** produced from dietary and metabolic sources (~50-100 mEq/day).
- For CO2-derived acid, the buffering occurs **intracellularly in RBCs** via hemoglobin before bicarbonate enters the plasma.
*Red blood cell bicarbonate*
- Bicarbonate is produced within RBCs from the dissociation of carbonic acid, but it is **not the buffer itself**.
- The bicarbonate is a **product** of the buffering reaction, not the buffering agent.
- Most RBC-produced HCO3- is transported to plasma via the **anion exchanger (Band 3 protein)** in exchange for Cl-.
*Plasma proteins*
- Plasma proteins like **albumin** have buffering capacity due to ionizable groups (imidazole groups of histidine residues).
- They contribute only about **1-5%** of total blood buffering capacity.
- Far less important than hemoglobin for buffering the large CO2-derived acid load.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Question 7: A pregnant woman is able to transfer oxygen to her fetus because fetal hemoglobin has a greater affinity for oxygen than does adult hemoglobin. Why is the affinity of fetal hemoglobin for oxygen higher?
- A. There is less 2,3-BPG in the fetal circulation as compared to maternal circulation
- B. Fetal hemoglobin binds 2,3-BPG with fewer ionic bonds than the adult form. (Correct Answer)
- C. The tense form of hemoglobin is more prevalent in the circulation of the fetus
- D. The oxygen-binding curve of fetal hemoglobin is shifted to the right.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Explanation: ***Fetal hemoglobin binds 2,3-BPG with fewer ionic bonds than the adult form.***
* **Fetal hemoglobin (HbF)**, composed of two alpha and two gamma subunits, interacts less effectively with **2,3-bisphosphoglycerate (2,3-BPG)** due to a difference in its gamma subunits compared to the beta subunits of **adult hemoglobin (HbA)**.
* The reduced binding of 2,3-BPG to HbF stabilizes its **R (relaxed) state**, which has a higher oxygen affinity, facilitating oxygen transfer from the mother to the fetus.
*There is less 2,3-BPG in the fetal circulation as compared to maternal circulation*
* While 2,3-BPG plays a crucial role in regulating oxygen affinity, the primary reason for **fetal hemoglobin's higher oxygen affinity** is its inherent structural difference that leads to weaker binding of 2,3-BPG, not necessarily the concentration of 2,3-BPG in the fetal circulation.
* The **concentration of 2,3-BPG is typically similar or even slightly higher in fetal blood** to enhance oxygen unloading at the tissues, but its effect on HbF is diminished.
*The tense form of hemoglobin is more prevalent in the circulation of the fetus*
* The **tense form (T-state)** of hemoglobin has a **lower affinity for oxygen**, and its prevalence would lead to reduced oxygen binding, which is contrary to the physiological need of the fetus to extract oxygen from the maternal blood.
* **Fetal hemoglobin's higher oxygen affinity** means it spends more time in the **relaxed form (R-state)**, which is responsible for tighter oxygen binding.
*The oxygen-binding curve of fetal hemoglobin is shifted to the right.*
* An **oxygen-binding curve shifted to the right** indicates a **decreased affinity for oxygen** and would facilitate oxygen unloading, not oxygen loading.
* For fetal hemoglobin to effectively extract oxygen from maternal blood, its **oxygen-binding curve must be shifted to the left**, signifying a higher oxygen affinity.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Question 8: Which porphyrin forms the organic component of heme?
- A. Uroporphyrin
- B. Coproporphyrin
- C. Deuteroporphyrin
- D. Protoporphyrin IX (Correct Answer)
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Explanation: ***Protoporphyrin IX***
- **Heme** is formed by the insertion of an **iron atom (Fe2+)** into the center of **protoporphyrin IX**.
- **Protoporphyrin IX** is the immediate precursor to heme in the **heme synthesis pathway**.
*Uroporphyrin*
- **Uroporphyrin** is an earlier precursor in the **heme synthesis pathway** and is much more hydrophilic than protoporphyrin.
- It accumulates in diseases like **congenital erythropoietic porphyria (CEP)**, leading to photosensitivity.
*Coproporphyrin*
- **Coproporphyrin** is an intermediate in the **heme synthesis pathway**, formed after uroporphyrinogen.
- It is also more water-soluble than protoporphyrin and its accumulation can be seen in various porphyrias.
*Deuteroporphyrin*
- **Deuteroporphyrin** is a synthetic porphyrin or a less common natural porphyrin that is not directly involved as the organic component of heme in mammals.
- While it is structurally similar to protoporphyrin, it does not serve as the direct precursor for heme formation in the human body.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Question 9: Why is blood stored in citrate-phosphate-dextrose considered more beneficial for hypoxic patients compared to blood stored in acidic-citrate-dextrose?
- A. The fall in 2,3-DPG is less. (Correct Answer)
- B. It has a higher pH level than acidic-citrate-dextrose.
- C. It is more effective in oxygen delivery.
- D. It has a longer shelf life than acidic-citrate-dextrose.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Explanation: ***The fall in 2,3-DPG is less.***
* **Citrate-phosphate-dextrose (CPD)** better preserves levels of **2,3-bisphosphoglycerate (2,3-DPG)** in stored red blood cells.
* Higher 2,3-DPG levels are crucial for **oxygen unloading** from hemoglobin in tissues, which is particularly beneficial for hypoxic patients who need efficient oxygen delivery.
*It has a higher pH level than acidic-citrate-dextrose.*
* While CPD does maintain a **less acidic pH** than acid-citrate-dextrose (ACD), which is generally favorable for red blood cell viability, the most direct benefit for hypoxic patients relates to 2,3-DPG.
* The slightly higher pH indirectly contributes to better 2,3-DPG preservation but isn't the primary reason for improved oxygen delivery.
*It is more effective in oxygen delivery.*
* While the *consequence* of using CPD is **more effective oxygen delivery** due to better 2,3-DPG preservation, this option describes the outcome rather than the underlying mechanism compared to the more specific answer regarding 2,3-DPG.
* The increased efficacy in oxygen delivery is directly attributable to the preserved 2,3-DPG levels.
*It has a longer shelf life than acidic-citrate-dextrose.*
* The storage solutions primarily impact red blood cell viability and function, but the **shelf life** (typically 21-35 days depending on the anticoagulant/preservative) is generally determined by other factors, including the additive solutions used with the anticoagulant.
* While CPD improves red blood cell quality, the primary advantage for hypoxic patients specifically lies in oxygen affinity rather than overall storage duration.
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG Question 10: Which factor has the most significant influence on the oxygen dissociation curve?
- A. 2,3-BPG (Correct Answer)
- B. pH
- C. Temperature
- D. All of these
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Explanation: ***2,3-BPG***
- **2,3-bisphosphoglycerate (2,3-BPG)** is a metabolic intermediate produced specifically in red blood cells that serves as the primary physiological regulator of hemoglobin's oxygen affinity.
- It binds to the central cavity of deoxygenated hemoglobin, stabilizing the tense (T) state and significantly decreasing oxygen affinity, shifting the curve to the right.
- Its concentration increases in chronic hypoxic conditions (high altitude, anemia, chronic lung disease), providing sustained adaptation for oxygen delivery to tissues.
- **2,3-BPG levels can increase by 50% or more** during chronic hypoxia, representing the most significant **long-term physiological mechanism** for modulating the oxygen dissociation curve.
*pH*
- A decrease in **pH** (Bohr effect) shifts the oxygen dissociation curve to the right by stabilizing the T state of hemoglobin.
- This is primarily an **acute response** to metabolic conditions rather than a sustained regulatory mechanism.
- While clinically important, pH changes are typically secondary to metabolic states rather than a primary regulatory mechanism.
*Temperature*
- An increase in **temperature** causes a rightward shift of the oxygen dissociation curve, promoting oxygen release from hemoglobin.
- Temperature effects are generally **passive responses** to environmental or metabolic conditions rather than active regulatory mechanisms.
- The magnitude of temperature-induced shifts is typically smaller than those produced by 2,3-BPG in physiological conditions.
*All of these*
- While pH, temperature, and 2,3-BPG all influence the oxygen dissociation curve, the question asks for the factor with the **most significant influence**.
- **2,3-BPG** is unique as the only factor that represents an **active, sustained, physiological regulatory mechanism** specifically evolved for oxygen delivery modulation.
- pH and temperature effects are important but represent **passive responses** to metabolic conditions rather than primary regulatory control mechanisms.
More Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve Indian Medical PG questions available in the OnCourse app. Practice MCQs, flashcards, and get detailed explanations.